 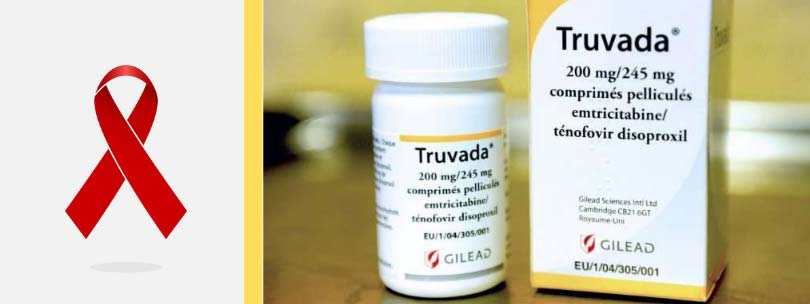
HIV/AIDS activists have been upset about the price of Truvada for PrEP for years. But it also has been criticized for excessive pricing in 2013, it sparked an uproar when it priced a 12-week course of the hepatitis C medication Sovaldi at $84,000.Ĭhristopher Rowland writes for the Washington Post. Gilead often works in cooperation with federal research agencies because it seeks to develop drugs to fight infectious diseases such as HIV/AIDS, hepatitis B and hepatitis C, epidemics that have attracted intense government attention. In addition to $50 million in government money, $17 million for the study came from the Bill & Melinda Gates Foundation, Grant said.īased on those clinical trials in humans, Gilead won approval to market Truvada for PrEP in 2012. Grant’s study, led from his lab in San Francisco and published in 2010 in the New England Journal of Medicine, proved that the drug lowered the risk of contracting HIV by more than 92%. The National Institutes of Health awarded grants to conduct more study, this time in clinical trials in humans. That began a nine-year effort, using two outside law firms retained by the CDC, which made their case to U.S. Gilead worked with the government, providing Truvada doses free of charge to test in the monkeys at CDC labs in Atlanta.Īfter Truvada was shown to work as a prophylactic in primates, the CDC applied for its patents. Gilead began marketing Truvada in 2004 after the Food and Drug Administration approved it to treat people who were already infected by HIV.Īt about the same time, government researchers at the CDC were looking for medicine that would do something to slow or end the epidemic: block HIV from infecting a healthy person, even if the person had unprotected sex. Trump’s newest budget, released this month, would add $291 million to fight the disease. 
To get close to that goal, specialists say, Truvada for PrEP needs to be far more widespread. President Trump set a goal in his State of the Union speech last month of eradicating HIV/AIDS by 2030. It would not discuss contents of the calls and letters, citing a confidentiality agreement with the CDC. “Since that time there have been several phone discussions and the exchange of about half a dozen letters addressing the parties’ concerns and positions,” Gilead said in a written statement in response to questions from the Washington Post. Gilead said it was contacted by the CDC about licensing Truvada for PrEP in 2016. The CDC itself did not respond to requests for comment. “When it comes to conversations with companies, those are subjects we’re not at liberty to talk about,” said Michael Mowatt, director of the office within the National Institute of Allergy and Infectious Diseases that represents the CDC in patent licensing matters. Government officials would not discuss steps they have taken to enforce the CDC patent. The case of Gilead and Truvada for PrEP adds a new twist to these debates, with demands that the government be more aggressive in exercising its own patent to ease the cost effects of a monopoly. Consumer advocates and members of Congress have stepped up demands that the government exercise its rights under existing law to license generic competition or imports during shortages or unwarranted price jumps several bills would enhance such “compulsory licensing” provisions. “They may not want to be in the position of suing these companies that arguably are producing valuable stuff,” he said.īut that stance increasingly is challenged by political anger over high drug prices. That tends to take patent infringement lawsuits off the table. NIH and CDC officials see their role as encouraging the commercialization of government-financed discoveries, not placing curbs on them, he said. Sukhatme, a professor and patent expert at Georgetown University’s law school. The government is not locked in a battle for profits the way a private company is, said Neel U. The fight over PrEP presents a novel challenge to the interdependence of drug companies and government developed over years of collaboration. Instead of enforcing its patent, Krellenstein said, the CDC is “twiddling their thumbs.” “The CDC has all these patents and is allowing Gilead to rip off the American people at the expense of public health,” said James Krellenstein, an HIV/AIDS activist and co-founder of the PrEP4All Collaboration, who has spent months digging into the government patents. Their complaints are part of a broader wave of anger over drug companies reaping hefty financial rewards by capitalizing on taxpayer-funded research. Activists want the government to take a more aggressive stance against Gilead. 
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
